 
Was unable to explain the scattering experiment of Rutherford.It did not mention anything about electron arrangement in the orbit.His model can’t explain hydrogen atomic spectra.Charged particles during acceleration would give out energy while revolving around nucleus it will lose energy and fall down into the nucleus.Although early atomic models were inaccurate and could not explain the results of some experiments, they served as the basis for future developments in quantum mechanics.He didn’t say anything about the electronic arrangement of atoms, which made his theory incomplete.It was not according to Maxwell’s principle and could not give an explanation for the steadiness of an atom.Deviation of alpha particles was because of positive charges in the foil.He discovered most of the atom was empty space.So, the quantity occupied by means of the positively charged particles in an atom may be very small in comparison to the total volume of an atom. Only a few of the α-debris have been deflected only a few α-particles had an almost 180 degree angle of deflection.The effective charge in an atom is concentrated to a completely small extent. Some of the α-debris have been deflected by using the gold sheet via very small angles, and subsequently the effective price in an atom is not uniformly disbursed.A chief fraction of the α-particles bombarded closer to the gold sheet surpassed through it without any deflection, and therefore most of the space in an atom is empty.Rutherford’s conclusion for the alpha scattering He made some observations that contradicted Thomson’s atomic model.To study the deformation created by alpha particles he placed a fluorescent zinc sulphide screen around a small gold plate.Direct the alpha energy from the radiation source to a thin sheet (100 nm thick) of gold. 
The trace of these particles was then studied after their contact with the gold plate.He did research by blasting a small sheet of gold with alpha particles.The Rutherford model shows that an atom is usually an empty space, with electrons surrounding a fixed nucleus, with positively charged in set, predictable pathways. 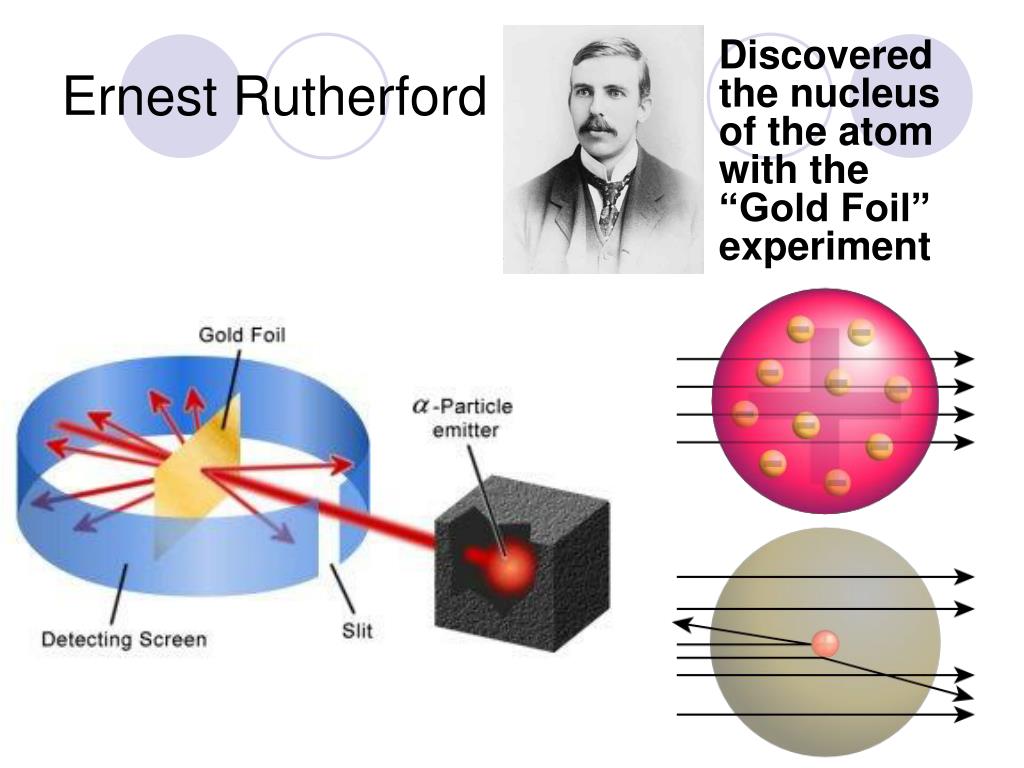
Ernest Rutherford, a British scientist who conducted the study and based on the observations of this study, proposed the atomic composition of the elements and presented the Rutherford Atomic Model. Thomson failed to explain certain experimental results associated with the atomic structure of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
